FORT WORTH, Texas — The Texas Second Court of Appeals in Fort Worth Thursday ruled a hospital can’t be forced to administer ivermectin to a COVID-19 patient.
According to court documents, 48-year-old Jason Jones, who is a Tarrant County law enforcement officer, was hospitalized after testing positive for COVID-19 in September. While he no longer has the virus, he suffered collapsed lungs and was placed in a medically induced coma and on a ventilator on Oct. 7.
Jones was hospitalized at Texas Health Hughley Hospital in Fort Worth.
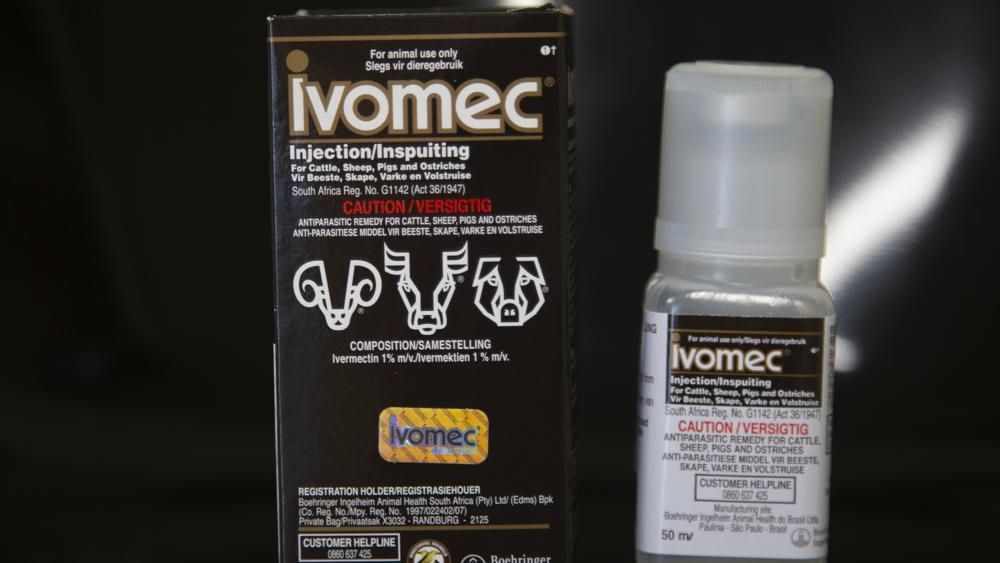
Court documents further state that Jones’ wife, Erin Jones, asked the hospital to administer ivermectin, which is typically used to eliminate parasitic worms, to her husband. She did so after consulting with Dr. Mary Talley Bowden, a physician who is not affiliated with the hospital and who had her privileges suspended by Houston Methodist Hospital after spreading false claims about COVID-19 on social media.
Dr. Bowden, who, among other things, touted ivermectin as a viable COVID-19 treatment and said approved vaccines are not effective, has since resigned from Houston Methodist.
Bowden prescribed the drug for Jason Jones, but Texas Health Hughely refused to administer it, prompting Erin Jones to file suit.
“But judges are not doctors. We are not empowered to decide whether a particular medication should be administered, or whether a particular doctor should be granted ICU privileges,” Chief Justice Bonnie Sudderth wrote in her opinion. “Our role is to interpret and apply the law as written. Although we may empathize with a wife’s desire to try anything and everything to save her husband, we are bound by the law, and the law in this case does not allow judicial intervention. Just as we cannot legislate from the bench, we cannot practice medicine from the bench. Therefore, we vacate the trial court’s temporary injunction.”
The U.S. Food and Drug Administration has approved ivermectin in both people and animals for some parasitic worms and for head lice and skin conditions. The FDA has not approved its use in treating or preventing COVID-19 in humans.
“Using any treatment for COVID-19 that’s not approved or authorized by the FDA, unless part of a clinical trial, can cause serious harm,” the FDA said in a warning about the drug.
The Associated Press contributed to this report.